Researchers Say Molybdenite Could Replace Silicon in Chips
Scientists at the Ecole Polytechnique Fédérale de Lausanne (EPFL) claim to have built an integrated circuit that uses molybdenite instead of silicon.
Get Tom's Hardware's best news and in-depth reviews, straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
The researchers believe that molybdenite can surpass the physical limits of silicon in terms of miniaturization, electricity consumption, and mechanical flexibility.
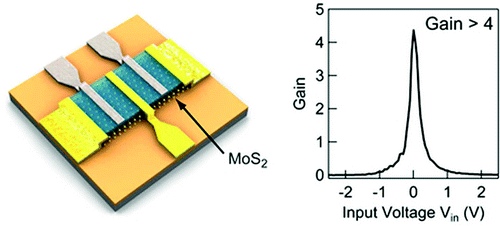
"The main advantage of MoS2 is that it allows us to reduce the size of transistors, and thus to further miniaturize them," said Andras Kis, director at the Laboratory of Nanoscale Electronics and Structures (LANES) at EPFL. "It has not been possible up to this point to make layers of silicon less than two nanometers thick, because of the risk of initiating a chemical reaction that would oxidize the surface and compromise its electronic properties. Molybdenite, on the other hand, can be worked in layers only three atoms thick, making it possible to build chips that are at least three times smaller. At this scale, the material is still very stable and conduction is easy to control."
What makes molybdenite even more attractive is the fact that it is a "relatively abundant, naturally occurring mineral." The researchers said that it can directly compete with silicon as its structure and semiconducting properties make it "an ideal material for transistors." According to Kis, however, molybdenite transistors are more efficient, since they "can be turned on and off much more quickly, and can be put into a more complete standby mode." The researcher said that the material is about as good as silicon for the amplification of electric signals.
Article continues belowSo far, the scientists have only built very basic chips with two to six transistors. They now want to build larger chips.
Get Tom's Hardware's best news and in-depth reviews, straight to your inbox.

Douglas Perry was a freelance writer for Tom's Hardware covering semiconductors, storage technology, quantum computing, and processor power delivery. He has authored several books and is currently an editor for The Oregonian/OregonLive.
-
alidan what happened to Graphene,was met with the silicon replacement, and of this new stuff have the same heat qualities that Graphene has, it be nice to see the chips going to 50 GHz+Reply -
dogman_1234 I was also thinking Graphene. Apparently, this is more efficient than Graphene. however, will this still be able to be used even under 16 nm nodes considering MoS2 is larger than SiO2 or even xC6,( Graphene)?Reply -
kcorp2003 According to Yu-Ming Lin from IBM Research:Reply
There is an important distinction between the graphene transistors that we demonstrated, and the transistors used in a CPU. Unlike silicon, ‘graphene does not have an energy gap, and therefore, graphene cannot be “switched off,” resulting in a small on/off ratio.
No off state means more energy leakage and therefore more heat. It also makes it more difficult to figure out the state of a transistor, and if IBM is correct, too difficult for it to be a viable solution when graphene is used as the base material.
-
cheepstuff graphene is also extremely expensive, but that is changing quickly and may no longer be true as new industrial processes are introducedReply -
memadmax nikorrIts all about money. The cheapest material wins...Reply
It will be expensive at first. When it takes off it will get cheaper... -
nikorrIts all about money. The cheapest material wins...Reply
"strawberry flavored cranberries"
Exactly... they even do it in your snack bars.